Summary
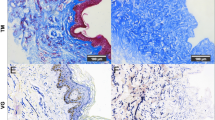
Many studies are being conducted to define the role of growth factors in cutaneous physiology in order to add cytokines in a timely fashion for optimal tissue engineering of skin. This study is aimed at developing a multistep approach for the production of bioengineered skin substitutes, taking into account the effects of various growth factors according to the culture time. The use of a serum-supplemented medium throughout the whole culture period of skin substitutes was compared to the sequential use of specific additives at defined culture steps. Histological analysis revealed that serum was necessary for keratinocyte proliferation and migration on dermal substitutes during the first 2 d after their seeding. However, the serum-free medium presented some advantages when supplemented with different additives at specific culture steps. Interestingly, ascorbic acid added to the dermal substitutes before and after keratinocyte seeding maintained their cuboïdal morphology in the basal epidermal layer. In the absence of serum, collagen matrix degradation slowed down, and a better multilayered epidermal organization was obtained, notably with retinoic acid. Stratum corneum formation was also enhanced by fatty acids. Thus, sequential addition of exogenous factors to the medium used to produce skin substitutes can improve their structural features and functional properties in vitro.
Similar content being viewed by others
References
Asselineau, D.; Bernard, B. A.; Bailly, C., et al. Retinoic acid improves epidermal morphogenesis. Dev. Biol. 133:322–335; 1989.
Asselineau, D.; Cavey, M.-T.; Shroot, B., et al. Control of epidermal differentiation by a retinoid analogue unable to bind to cytosolic retinoic acid-binding proteins (CRABP). J. Invest. Dermatol. 98:128–134; 1992.
Auger, F. A. The role of cultured autologous human epithelium in large burn wound treatment. Transplantation/Implantation Today 5:21–24; 1988.
Auger, F. A.; Guignard, R.; López Valle, C. A., et al. Role and inocuity of Tisseel®, a tissue glue, in the grafting process and in vivo evolution of human cultured epidermis. Br. J. Plast. Surg. 46:136–142; 1992.
Auger, F. A.; López Valle, C. A.; Guignard, R., et al. Skin equivalents produced using human collagens. In Vitro Cell. Dev. Biol. 31:432–439; 1995.
Bailly, C.; Drèza, S.; Asselineau, D., et al. Retinoic acid inhibits the production of collagenase by human epidermal keratinocytes. J. Invest. Dermatol. 94:47–51; 1990.
Bell, E.; Ehrlich, H. P.; Buttle, D. J., et al. Living tissue formed in vitro and accepted as skin-equivalent tissue of full thickness. Science 211: 1052–1054; 1981a.
Bell, E.; Ehrlich, H. P.; Sher, S., et al. Development and use of a living skin equivalent. Plast. Reconstr. Surg. 67:386–392; 1981b.
Bell, E.; Ivarsson, B.; Merrill, C. Production of a tissue-like structure by contraction of collagen lattices by human fibroblasts of different proliferative potential in vitro. Proc. Natl. Acad. Sci. USA 76:1274–1278; 1979.
Berthod, F.; Germain, L.; Guignard, R., et al. Differential expression of collagens XII and XIV in human skin and in reconstructed skin. J. Invest. Dermatol. 108:737–742; 1997.
Black, A.; Berthod, F.; L'Heureux, H., et al. In vitro reconstruction of a human capillary-like network in a tissue engineered skin equivalent. FASEB J. 12:1331–1340; 1998.
Bligh, E. G.; Dyer, W. J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37:911–917; 1959.
Boisseau, A. M.; Donatien, P.; Surleve-Bazeille, J. E., et al. Production of epidermal sheets in a serum free culture system: a further appraisal of the role of extracellular calcium. J. Dermatol. Sci. 3:111–120; 1992.
Bouvard, V.; Germain, L.; Rompré, P., et al. Influence of dermal equivalent maturation on a skin equivalent development. Biochem. Cell. Biol. 70:34–42; 1992.
Boyce, S. T.; Foreman, T. J.; English, K. B., et al. Skin wound closure in athymic mice with cultured human cells, biopolymers, and growth factors. Surgery 110:866–876; 1991.
Boyce, S. T.; Medrano, E. E.; Abdel-Malek, Z., et al. Pigmentation and inhibition of wound contraction by cultured skin substitutes with adult melanocytes after transplantation to athymic mice. J. Invest. Dermatol. 100:360–365; 1993.
Boyce, S. T.; Williams, M. L. Lipid supplemented medium induces lamellar bodies and precursors of barrier lipids in cultured analogues of human skin. J. Invest. Dermatol. 101:180–184; 1993.
Choi, Y.; Fuchs, E. TGF-β and retinoic acid: regulators of growth and modifiers of differentiation in human epidermal cells. Cell (Regul.) 1:791–809; 1990.
Contard, P.; Bartel, R. L.; Jacobs II, L., et al. Culturing keratinocytes and fibroblasts in a three-dimensional mesh results in epidermal differentiation and formation of a basal lamina-anchoring zone. J. Invest. Dermatol. 100:35–39; 1993.
Coulomb, B.; Dubertret, L.; Merrill, C., et al. The collagen lattice: a model for studying the physiology, biosynthesis function and pharmacology of the skin. Br. J. Dermatol. 111:83–87; 1984.
Coulomb, B.; Friteau, L.; Baruch, J., et al. Advantage of the presence of living dermal fibroblasts within in vitro reconstructed skin for grafting in humans. Plast. Reconstr. Surg. 101:1891–1903; 1998.
Coulomb, B.; Lebreton, C.; Dubertret, L. Influence of human dermal fibroblasts on epidermalization. J. Invest. Dermatol. 92:122–125; 1989.
Eaglstein, W. H.; Falanga, V. Tissue engineering and the development of Apligraf, a human skin equivalent. Clin. Therap. 19:894–905; 1997.
Fowler, S. D.; Greenspan, P. Application of Nile red, a fluorescent hydrophobic probe, for the detection of neutral lipid deposits in tissue sections: comparison with oil red O. J. Histochem. Cytochem. 33: 833–836; 1985.
Gallico, G. G.; O'Connor, N. E.; Compton, C. C., et al. Permanent coverage of large burn wounds with autologous cultured human epithelium. N. Engl. J. Med. 331:448–451; 1984.
Geesin, J. C.; Brown, L. J.; Gordon, J. S., et al. Regulation of collagen synthesis in human dermal fibroblasts in contracted gels by ascorbic acid, growth factors, and inhibitors of lipids peroxidation. Exp. Cell Res. 206:283–290; 1993.
Germain, L.; Rouabhia, M.; Guignard, R., et al. Improvement of human keratinocyte isolation and culture using thermolysin. Burns 19:99–104; 1993.
Green, H.; Kehinde, O.; Thomas, J. Growth of cultured human epidermal cells into multiple epithelia suitable for grafting. Proc. Natl. Acad. Sci. USA 76:5665–5668; 1979.
Hansbrough, J. F.; Boyce, S. T.; Cooper, M. L., et al. Burn wound closure with cultured autologous keratinocytes and fibroblasts attached to a collagen-glycosaminoglycan substrate. JAMA 262:2125–2130; 1989.
Hashiro, M.; Matsumoto, K.; Hashimoto, K., et al. Stimulation of fibronectin secretion in cultured human keratinocytes by transforming growth factor-β not by other growth inhibitory substances. J. Dermatol. 18: 252–257; 1991.
Jetten, M. A. Multi-stage program of differentiation in human epidermal keratinocytes: regulation by retinoids. J. Invest. Dermatol. 95:44s-46s; 1990.
Kratz, G.; Haegerstrand, A.; Dalsgaard, C.-J. Conditioned medium from cultured human keratinocytes has growth stimulatory properties on different human cell types. J. Invest. Dermatol. 97:1039–1043; 1991.
López Valle, C. A.; Auger, F. A.; Rompré, P., et al. Peripheral anchorage of dermal equivalents. Br. J. Dermatol. 127:365–371; 1992.
López Valle, C. A.; Germain, L.; Rouabhia, M., et al. Grafting on nude mice of living skin equivalents produced using human collagens. Transplantation 62:317–323; 1996.
López Valle, C. A.; Glaude, P.; Auger, F. A., et al. Tie-over dressings: surgical model for in vivo evaluation of cultured epidermal sheets in mice. Plast. Reconstr. Surg. 89:139–143; 1992.
Matsue, H.; Cruz, P. D.; Bergstresser, P. R., et al. Cytokine expression by epidermal cell subpopulations. J. Invest. Dermatol. 99:42S-45S; 1992.
Michel, M.; Auger, F. A.; Germain, L. Anchored skin equivalent cultured in vitro: a new tool for percutaneous absorption studies. In Vitro Cell. Dev. Biol. 29A:834–837; 1993.
Michel, M.; L'Heureux, N.; Auger, F. A., et al. From newborn to adult: phenotypic and functional properties of skin equivalent and human skin as a function of donor age. J. Cell. Physiol. 171:179–189; 1997.
Nolte, C. J. M.; Oleson, M. A.; Parenteau, N. L. Development of a stratum corneum and barrier function in an organotypic skin culture. Arch. Dermatol. Res. 285:466–474; 1993.
Oikarinen, A.; Kylmäniemi, M.; Autio-Harmainen, H., et al. Demonstration of 72-kDa and 92-kDa forms of type IV collagenase in human skin: variable expression in various blistering diseases, induction during re-epithelialization, and decrease by topical glucocorticoïds. J. Invest. Dermatol. 101:205–210; 1993.
Phillips, C. L.; Tajna, S.; Pinnell, S. R. Ascorbic acid and transforming growth factor-β increase collagen biosynthesis via different mechanisms: coordinate regulation of pro-a(I) and pro-a(III) collagens. Arch. Biochem. Biophys. 295:397–403; 1992.
Ponec, M. Reconstruction of human epidermis on de-epidermized dermis: expression of differentiation-specific protein markers and lipid composition. Toxicol. In Vitro 5:597–606; 1991.
Ponec, M.; Weerheim, A., et al. Retinoids and lipid changes in keratinocytes. Methods Enzymol. 190:30–41; 1990.
Ponec, M.; Weerheim, A.; Kempenaar, J., et al. Lipid composition of cultured human keratinocytes in relation to their differentiation. J. Lipid Res. 29:949–961; 1988.
Pruitt, B. A. The evolutionary development of biologic dressings and skin substitutes. J. Burn Care Rehabil. 18:S2-S5; 1997.
Rheinwald, J. G.; Green, H. Serial cultivation of strains of human epidermal keratinocytes: the formation of keratinizing colonies from single cells. Cell 6:331–343; 1975.
Rompré, P.; Auger, F. A.; Germain, L., et al. The Influence of initial collagen and cellular concentrations on the final surface area of dermal and skin equivalents: a Box-Behnken analysis. In Vitro Cell. Dev. Biol. 26:983–990; 1990.
Sanquer, S.; Mark, S. E.; Gilchrest, B. A. Retinoids and state of differentiation modulate CRABP II gene expression in skin equivalent. J. Invest. Dermatol. 100:148–153; 1993.
Shakespeare, V.; Shakespeare, P. Effects of granulation-tissue-conditioned medium on the growth of human keratinocytes in vitro. Br. J. Plast. Surg. 44:219–223; 1991.
Tinois, E.; Tiollier, J.; Gaucherand, M., et al. In vitro and post-transplantation differentiation of human keratinocytes grown on the human Type IV collagen film of a bilayered dermal substitute. Exp. Cell Res. 193: 310–319; 1991.
Varani, J.; Fligiel, S. E.; Schuger, L., et al. Effects of all-trans retinoic acid and calcium on human skin in organ culture. Am. J. Pathol. 1421: 189–198; 1993.
Varani, J.; Inman, D. R.; Gibbs, D. F., et al. Modulation of calcium levels in keratinocytes by all-trans retinoic acid. Pathobiology 60:93–99; 1992.
Vicanova, J.; Boyce, S. T.; Harriger, M. D., et al. Stratum corneum lipid composition and structure in cultured skin substitutes is restored to normal after grafting onto athymic mice. J. Invest. Dermatol. 3:114–120; 1998.
Wagnaldo, T.; Bernard, F.; Asselineau, D., et al. Expression of loricrin is negatively controlled by retinoid acid in human epidermis reconstructed in vitro. Differentiation 49:39–46; 1992.
Xu, W.; Germain, L.; Goulet, F., et al. Permanent grafting of living skin substitutes: surgical parameters to control for successful results. J. Burn Care Rehabil. 17:7–13; 1996.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Auger, F.A., Pouliot, R., Tremblay, N. et al. Multistep production of bioengineered skin substitutes: Sequential modulation of culture conditions. In Vitro Cell.Dev.Biol.-Animal 36, 96–103 (2000). https://doi.org/10.1290/1071-2690(2000)036<0096:MPOBSS>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1071-2690(2000)036<0096:MPOBSS>2.0.CO;2